| Course of LABORATORY MEDICINE Disturbances of the endocrine system Hormones are produced by endocrine glands and serve to regulate the physiological functions of many organs and tissues. Their concentration in the blood is usually finely regulated thus their concentration and its circadiane changes vary within a limited range. Disregulation may occur both in the sense of excess secretion (often due to benign, hormone-secreting tumours of the gland) or in the sense of insufficient secretion (possibly due to damage of the gland due to metabolic defects, malignancies, insufficient stimulation). Moreover, alterations of the circadian rythms of secretion occur and in some instances cause symptoms and have diagnostic value. From a biochemical standpoint, hormones belong to several different families of compounds, and every endocrine gland may secrete one or more hormones, usually belonging to the same biochemical family. An exception is the thyroid which secretes two hormones belonging to different biochemical families: Thyroxin is an aminoacid derivative, whereas Thyrocalcitonin is a small protein. The adrenals do secrete hormones of two very different chemical types (catecholamines are aminocid derivatives, corticoids are cholesterol derivatives), but form an embriological viewpoint they are two different glands one of which encircles the other. As a consequence of this variety several analytical methods are required, as summarized in the Table below:
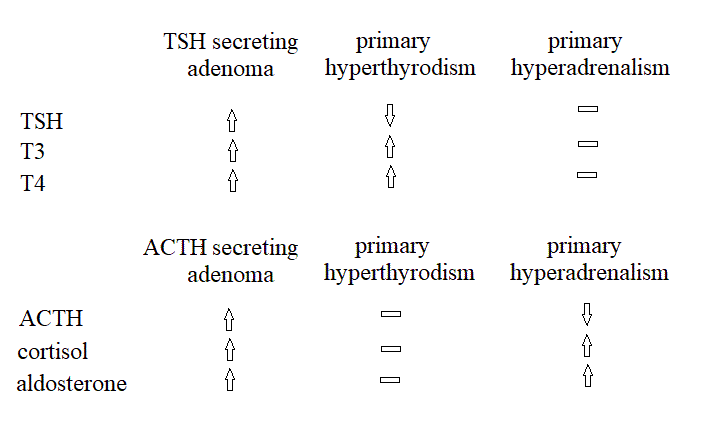
Disorders of the anterior pituitary. Hypopituitarism can be selective or generalized (panhypopituitarism). Generalized hypoituitarism is the result of compressive or ischemic destruction of the hypophysis, often due to tumours; a less common condition is the Sheehan syndrome (ischemic necrosis of the hypophysis due to post partum hemorrages and shock). Gonadotropins are lost first, followed by GH, and then by TSH and ACTH. Death may ensue, due to the lack of thyroid and adrenal stimulation. Diagnosis is by imaging methods (lesions of the sella turcica) and by laboratory determination of the hormones' concentration in the serum. Infertility and a syndrome indistinguishable from Addison's disease characterize the clinical picture. An important differential diagnosis is with anorexia nervosa. Diagnosis is confirmed by the measurement of the concentration of pituitary hormones, and of the hormones they stimulate (T3, T4, cortisol, etc.) that result low. Selective hypopituitarism, with isolated pituitary deficiencies, is suspected because of failure to grow (pituitary nanism due to deficiency of GH), or because of hypothyrodism or adrenal insufficiency (pituitary Addison's disease), or sterility and lack of menstruations. Hyperpituitarism, i.e. the pathological increase of pituitary function, is usually selective and may be idiopathic or due to a benign neoplasia of the gland that secretes one of its hormones. In the case of GH the syndrome causes pituitary gigantism (in the child) or acromegaly (in the adult). Increased secretion of ACTH leads to the pituitary form of the Cushing disease, and the symptoms are due to the increased production of cortisol. Disorders of the posterior pituitary. Diabetes insipidus is due to insufficient or absent production of ADH. Since this hormone causes the kidney to reabsorb water, its deficiency is associated with poliuria and diluted urine (to be distinguished from diabetic mellituria, in which the urine flow is increased because of the osmotic effect of glucose, and from nephrogenic diabetes insipidus, in which the kidney's function is reduced). The pars intermedia of the hypophysis is active in the foetus but very small or absent in the human adult (it remains functional in lower vertebrates, and may be responsible for skin colour changes linked to camouflage). In the foetus it produces the Melanocyte Stimulating Hormone (MSH). Is not responsible of diseases in humans. Disorders of the thyroid. Hypothyroidism may be due to lack of iodine in the diet, or to inflammatory diseases of the gland. The thyroid is usually enlarged (goiter) and the concentration of T4 and T3 in the blood is decreased. The clinical picture is ususally quite characteristic and diagnosis is confirmed by laboratory findings. If iodine administration does not cure the syndrome a substitutive therapy is started. Hyperthyroidism (Basedow's disease; Graves' disease; thyrotoxicosis) may be due to diffuse enlargement of the gland (in some causes because of a benign adenoma of the pituitary, which secretes TSH) or to a secreting adenoma. 131I scintigraphy is diagnostic, and the laboratory analyses will confirm increased serum concentration of T3 and T4 (and possibly of TSH). Surgery is indicated. Disorders of the adrenal cortex. Adrenal insufficiency (Addison's disease). The symptoms of Addison's disease are dominated by the reduced production of glucocorticoid hormones. The disease is fatal if untreated. The major causes of the destruction of adrenal cortex are: tuberculosis, cancer, amiloidosis and inflammatory necrosis. A clinically similar condition may be due to panhypopituitarism and by the (uncommon) selective reduction of hypophisary secretion of ACTH. Addison's disease due to the destruction of the adrenal cortex is characterized by increased serum ACTH; that due to panhypopitutarism by decreased or absent serum ACTH. Glucocorticoid hypersecretion (Cushing's disease) may be due to a functioning adenoma of the hypophysis (that secretes ACTH) or of the adrenal cortex (that secretes cortisol). Less frequently, tumors of other tissues (e.g. the lung) can produce ACTH. In typical cases of Cushing's disease, the secretion of both glucocorticoid and mineralocorticoid hormones is incerased. Cortisol secretion follows a circadian rhythm being higher in the morning (serum concentration 9-24 μg/dL) and lower in the afternoon (3-12 μg/dL); thus the measured concentration should be referred to the time of sampling. Alternatively the concentration can be measured in the urines collected over the 24 hours (cortisol: 20-100 μg/24 hours; cortisol metabolytes - 17 hydroxycorticoid derivatives: 2-10 mg/24 hours). An important diagnostic test is the dexamethasone suppression test. 0.5 mg of dexamethasone every 6 hours should strongly depress the production of ACTH and hence that of cortisol. Adenomas of the adrenal usually do not respond to dexamethasone suppression. Hyperaldosteronism. Aldosterone can be measured in the plasma (normal value 1-5 ng/dL) or in the urine (2-10 μg/24 hours). Sex hormones are produced primarily by the gonads, but also by the adrenal cortex. As a general rule, their secretion is estimated from the metabolytes (17 ketosteroid derivatives) in the urine (normal values: make 7-25 mg/24 hours; female 4-15 mg/24 hours). Home of this course Slides for this lecture:
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||